ALL Q-Fusion Screening Kit
A novel, multiplex system for simultaneous detection of 16 fusion genes in leukemia patients.
The ability to detect cancer specific fusion genes for Acute Lymphoblastic Leukemia (ALL) can ensure optimal treatment selection and appropriate disease monitoring. While detection of fusion genes is critical in both research and clinical settings, current methodologies to detect such fusion genes have a number of shortcomings.
QuanDx’s ALL Q-Fusion Screening Kit offers a novel methodology for the detection of ALL fusion genes, providing rapid results for a fraction of the cost of traditional methodologies like FISH. In addition to being faster and more cost effective than other methods of detection, the ALL Q-Fusion Screening Kit, which relies on QuanDx’s proprietary Yin-Yang probe technology, reduces the complexity involved with testing and can be easily performed for routine screening.
Key Features:
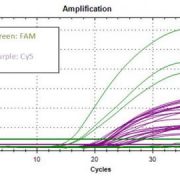
- Multiplexing: Allows simultaneous detection of 16 fusion genes with 71 breakpoints.
- Compatibility: Compatible with most current real-time qPCR instruments.
- Rapid: Results can be obtained in 2-4 hours.
- Simple: Easy to perform for routine screening.
- Cost Effective: Can be performed for a fraction of the cost of FISH.
For Research Use Only. Not intended for use in diagnostic procedures
| Fusion genes | MLL-AF4, TEL-AML1, MLL-ENL, E2A-PBX1, BCR-ABL P210, BCR-ABL P190, SIL-TAL1, E2A-HLF, MLL-AF6, CALM-AF10, HOX11, HOX11L2, MLL-AF10, SET-CAN, TEL-ABL1, TLS-ERG, NPM-ALK |
|---|---|
| Sample types | Bone marrow, peripheral blood, FFPE, and cell lines |
| Reaction Volume | 25 ul |
| Reaction time | 2-3 hour |
| Internal control | GUSB as housekeeping genes, 2 positive control, 3 negative control |
| Sensitivity | 10-4 |
| Instruments | Compatible with Bio-Rad CFX96, iCycler, ABI7500, Stratagene Mx3000P/Mx3005P, and other commonly used RT-qPCR machines. |
Product Flyer
| Cat. No. | Description | Size |
|---|---|---|
| 1-100-30010 | ALL Q-Fusion Screening Kit |
10-reactions |